- Author: Siavash Taravati
Western drywood termites (Incisitermes minor, Figure 1) are important pests of structural wood in California, causing millions of dollars in damage annually. These termites are very cryptic, hidden in their galleries within wood members (pieces of wood), and only emerge during swarming. As a result, wood damage usually goes unnoticed for a long time.
UC IPM Control options are generally categorized as either whole-structure treatment (heat-treatment and fumigation) or local treatments (insecticide injection into the wood, high-power microwaves, electrocution, and other techniques). Despite the high efficacy of fumigation, there has been increasing interest by property owners to use local treatments for eradicating drywood termites. This may be due to the high cost and inconvenience of fumigation.
To learn more about decision-making associated with fumigation, visit urbanipmsocal.com/ipm/termites/ to-fumigate-or-not-to-fumigate. Local treatment of drywood termites can be ineffective because of the difficulty in locating active infestation sites within structures. To address this issue, practitioners and researchers have considered different detection methods using traditional and modern technologies such as borescopes, moisture meters, and heat sensors as well as devices using X-rays, acoustic emission, and low-energy microwaves.
Here we provide a review and some technical details on how to operate a specific device using microwave technology for detecting termite movement in structures.
Termatrac
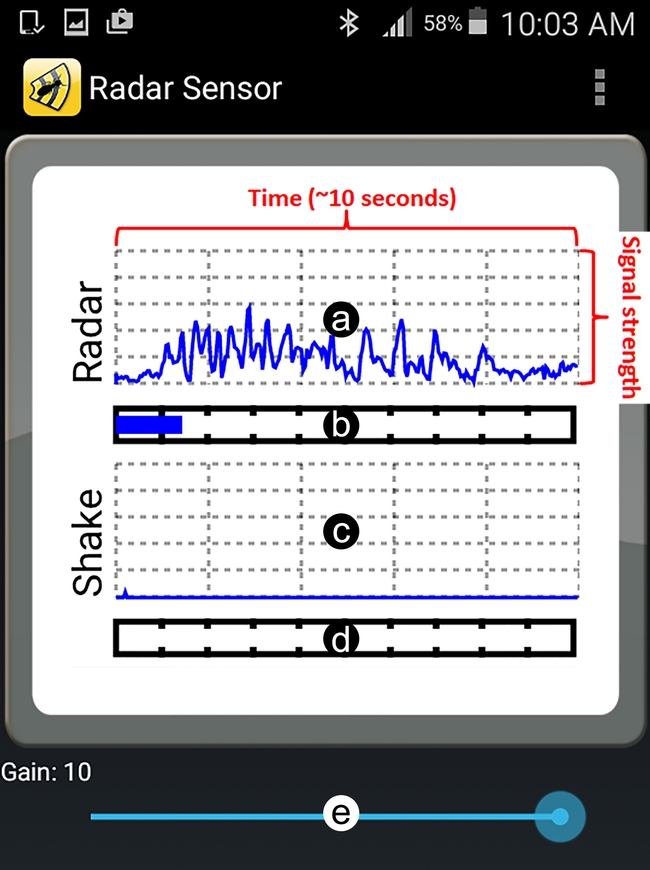
Termatrac1 is the brand name of a portable device that emits and receives low-energy microwaves to detect tiny movements in wood. This device is currently available as two models: Termatrac T3i All Sensor and Termatrac T3i Radar Only. Termatrac All Sensor includes a microwave emitter/sensor, a moisture meter, and a thermal sensor. The Radar-Only version, however, includes only the microwave technology (“radar”). Both devices generate a line graph output that represents termite movement within wood
Although such output can be informative, interpreting the results might not always be easy and may also require considerable expertise. First, the output's line graph may represent detection of non-termite objects or the user themselves (body movement or hand shaking while holding the device). Second, the signal intensity varies depending on the depth of termite activity, so Gain settings may need to be adjusted for higher or lower sensitivity. Third, the relationship between termite density (number of termites per unit of area) and signal strength is not easily understood by users (Figure 3). Fourth, termites may not be present or active during inspection and this may lead to a false negative conclusion (concluding “no termites” when they are present) when inspecting an infestation. To address these issues, field and lab research experiments were conducted in California to evaluate the efficacy of the Termatrac device and to help termite inspectors accurately interpret the output signal.
Termatrac can be used in different positions (see Figure 4):
- hand-held with radar surface flush against the inspection surface
- mounted on a tripod with radar surface flush against the inspection surface
- resting on a horizontal surface with radar surface flush against the inspection surface
- with radar at 45° angle to the inspection surface using the back flap or a tripod
Field studies revealed that hand-held uses produce less accurate results than tripod/flap supported uses due to user hand shaking. Also, the device's output showed more noise (noise refers to a detected signal in the output that is not coming from drywood termites) from the user's body movement when used at 45° to the inspection surface as compared to flush against the inspection surface (Taravati 2019).
Recommendations
For optimal readings, Termatrac users should keep the following in mind. Users need to stand still when reading the output or the device will pick up their body movement and produce a false positive signal. This is especially true at high sensitivities. Users also need to ensure that there are no moving objects (vehicles, plants swaying with the wind, airborne debris such as leaves and dusts, children, or animals (such as pets and birds) on the other side of the inspection surface (a wall for instance) which may create false positive signals. Also, water passing through pipes behind inspection surfaces may produce a strong signal. However, heavy machinery around the experiment sites did not produce any detectable noise despite being very loud. The device should not be used to inspect unstable surfaces or non-fixed objects (e.g. yard fence) since these situations will increase the chance of false positive signals and inaccurate detection of termites. To save time and increase accuracy when inspecting standard interior walls, users should first try to locate studs using a stud finder and then use Termatrac on those areas only. Users may also choose to focus on wooden window frames and windowsills since these have been observed to be one of the most common spots where drywood termites are detected in homes.
Findings
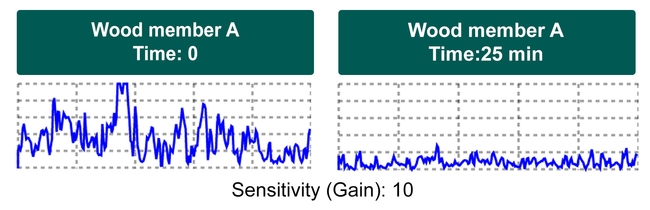
Lab studies showed that higher densities of termites may not necessarily produce stronger signal (Figure 3).
At the highest sensitivity setting, Termatrac T3i was able to detect a single drywood termite behind 5 cm (2 inch) of wood and 1.3 cm (0.5 inch) of drywall (total thickness of test “wall”: 6.3 cm / 2.5 inch). Drywood termites move within their galleries continually and therefore may not be present in all gallery regions at all times. Furthermore, termite activity may change throughout the day depending on temperature and other factors (Figure 5). As a result, if you suspect an active infestation in a wall but are not getting a detectable Termatrac signal, it is worth moving on to other areas and then returning in a few minutes to reinspect the suspect location.
This structural wood member contained 47 live termites. (from Taravati, 2019) To conclude, Termatrac can be very useful in some termite detection. Like other termite detection devices, Termatrac has limitations and requires training and experience before a user can efficiently and accurately detect termites. With this said, an experienced Termatrac user can obtain valuable information about termite presence and activity when the infested wood members are in accessible locations.
1 Mention of a product does not constitute an endorsement.
This article was originally published in Winter 2020 issue of The Green Bulletin.

- Author: Cheryl Reynolds
- Author: Petr Kosina

Cilantro and parsley growers have something else to be happy about: The UC Statewide IPM Program just released new Pest Management Guidelines for Cilantro and Parsley.
Cilantro and parsley are herbs used both fresh and dry for preparation of many popular dishes in almost all parts of the world including California. Apart from their pleasant flavor, both plants are also known for a number of nutritional and health benefits.
In California, cilantro and parsley are grown commercially on more than 7,000 acres, primarily along the southern and central coast. Cilantro (also known as Chinese or Mexican parsley) and parsley are examples of specialty vegetable crops important in crop rotations and in contributing to California's overall agricultural diversity.


Check out the new guidelines and other pest management information on the UC IPM website.
- Author: Kathy Keatley Garvey

“To say that Dr. Mary Louise Flint has been key to the success of California agriculture is not a misstatement or exaggeration,” Borg told the attendees. “The greatest threats to California's agriculture are the myriad of introduced and native pests and diseases. Keeping agricultural pests and diseases at bay while preserving the environment by an informed, balanced and interdisciplinary approach, an approach that is a hallmark of UC Davis.”

Borg, who chaired the Meyer Award Committee and served as the emcee at the dinner, noted that Flint was part of the original team that established the UC IPM Program in 1980. Since then her passion and vision for ecologically based IPM has influenced almost every aspect of IPM in the state, he said.
Before introducing her, Borg said: “I want to put her work in the context that it deserves and to do that, we need to understand a little about the significance of California agriculture.”
“In 2012 California was the top agricultural state in cash receipts at $44.7 billion. Iowa was second at $31.9 billion. Using data from 2007, Paul F. Starrs and Peter Goin in their Field Guide to Agriculture (UC Press 2010) make the case, for $37 billion in cash receipts for California's crops represents an agricultural economy of between $200-$300 billion.”

Borg related that when he “first started as an agriculture librarian over 30 years ago, I was introduced to Mary Louise Flint's work in IPM.” He praised her legendary status in California agriculture, “especially with regard to the University of California's role in California agriculture.”
He read a portion of the nomination letter by Michael Parrella, professor and chair of the UC Davis Department of Entomology and Nematology, that said: “Her name is synonymous with IPM, pest control alternatives, and public service, not just in California and the United States, but worldwide.” Parrella also noted that Flint “has been heavily involved in the leadership, creativity and success of UC IPM Program since 1982 and is UC IPM's longest-tenured employee.”


The UC Davis entomologist traced her history with the organization. “I remember attending the California State Fair in the early 1980s and being somewhat horrified to find the UC Master Gardeners answering questions using the Ortho Problem Solver and the Rodale Guide to Organic Gardening,” she said. “I was told that they relied on these books because UC provided very little garden pest management information.” That helped motivate her to write “Pest of the Garden and Small Farm.” From there, she and her colleagues moved on to Pest Management Guidelines, Pest Notes, Quick Tips, UC IPM website, UC IPM kiosk, our YouTube channel, blog and Twitter.
Flint is the third entomologist (Frank Zalom, 2004, and Thomas Leigh, 1988) to receive the Academic Federation award, first presented in 1971.